Biologics, mRNA therapies, antibody drug conjugates (ADCs), and other next-generation pharmaceuticals are highly effective but also extremely sensitive. Ensuring their stability requires more than suitable primary packaging—it demands intelligent secondary solutions that safeguard against light, oxygen, and extreme temperatures. At Schreiner MediPharm, Corinna Endres, Product Manager Drug Protection, is responsible for label systems designed specifically for these challenges. In this interview, she highlights current demands and emerging innovations in drug protection.
“Modern APIs react extremely sensitively—packaging them is a true balancing act.”
Corinna, what challenges do modern active ingredients (APIs) such as biologics or mRNA drugs pose for packaging?
Many of these APIs are highly reactive to external factors such as light, oxygen, and temperature. That makes their packaging particularly complex. Transparent containers—such as COC syringes—offer advantages, but their lower oxygen barrier means additional protection is essential. The goal is always to maintain drug stability and effectiveness without compromising container functionality or required visual inspection. With today’s ultra-sensitive drugs, finding that balance is often a major challenge.
Light Protection: Modular Solutions for Every Sensitivity Level
One of your key focus areas is light protection. What solutions do you offer?
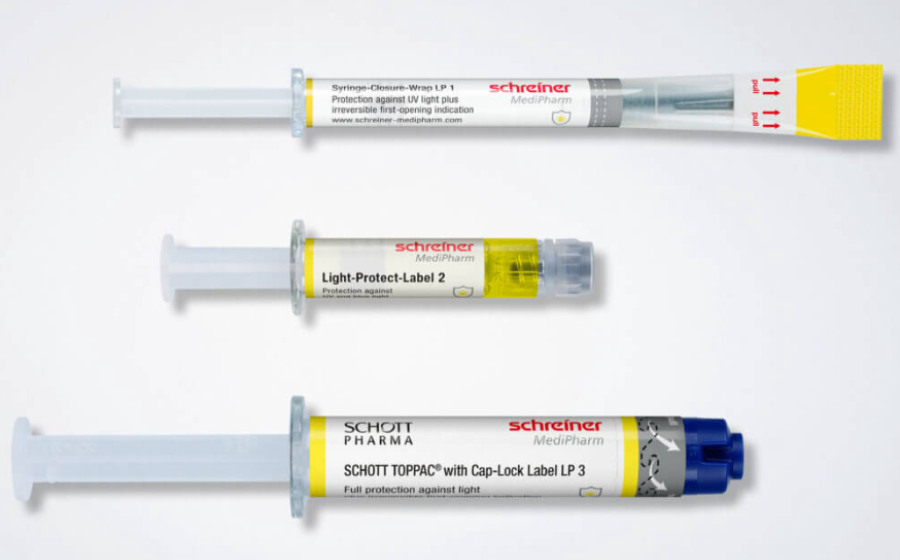
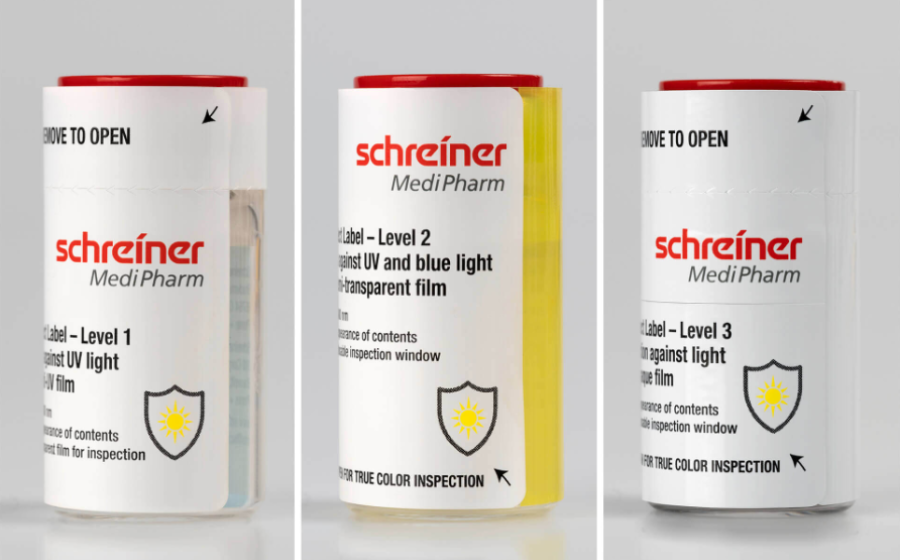
Our light-protection solutions follow a modular concept tailored to the drug’s sensitivity. We offer three protection levels—from UV protection with a transparent inspection window to full light protection with a resealable window. These options can be integrated into different label formats.
- Light-Protect-Label – Provides customized UV and light protection for vials and syringes.
- Syringe-Closure-Wrap Label – Wraps around the syringe barrel and cap and includes a first-opening indication.
- Cap-Lock Label – Designed for larger syringes; encloses both the barrel and cap while adding a sealing function.
Temperature Stability: For Deep-Frozen mRNA and Advanced Therapies
What about temperature-related requirements—for instance, for mRNA drugs?
Deep-freezing has become increasingly important. Many mRNA, cell, or gene-based drugs must be stored at –80 °C or even lower. For these conditions, we developed the Freeze-Light-Protect Label, which adheres reliably even at extreme sub-zero temperatures. It can be combined with different light-protection levels and additional features such as graduations or documentation components.
Gas Barrier Protection: Reducing Oxygen Diffusion
And how can protection against oxygen be enhanced?
We provide labels with integrated gas-barrier layers that significantly reduce oxygen diffusion—an issue especially relevant for COC syringes. Depending on requirements, we offer two variants:
- A large-surface barrier label covering most of the syringe barrel
- The Syringe-Closure-Wrap version, which also encloses the cap and includes an irreversible first-opening indication for added tamper evidence




