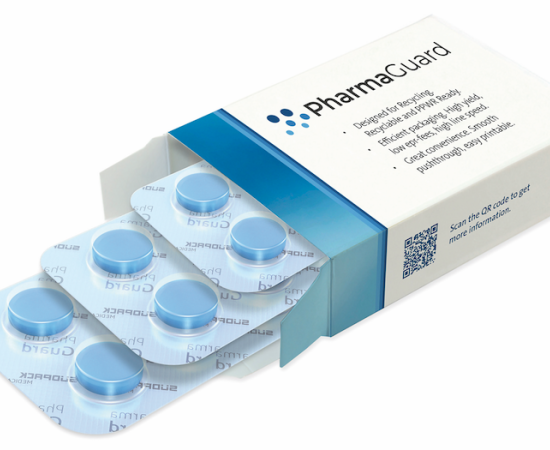
In a significant move toward advancing pharmaceutical packaging innovation and sustainability, Honeywell (NASDAQ: HON) has announced that Evertis, a prominent film producer, has selected its medical-grade Aclar® film for use in the Evercare brand’s pharmaceutical applications. This collaboration aims to ensure safer, longer-lasting medication packaging while promoting recyclable solutions within the pharmaceutical industry.
- Today is: