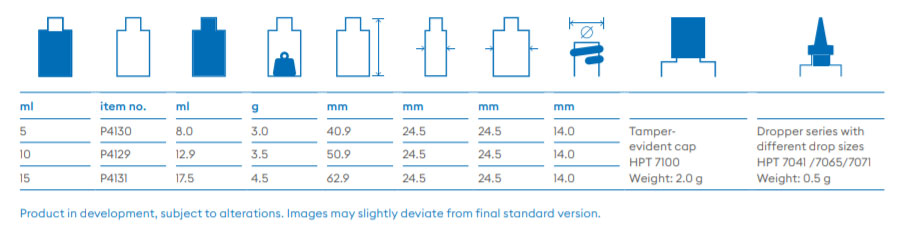
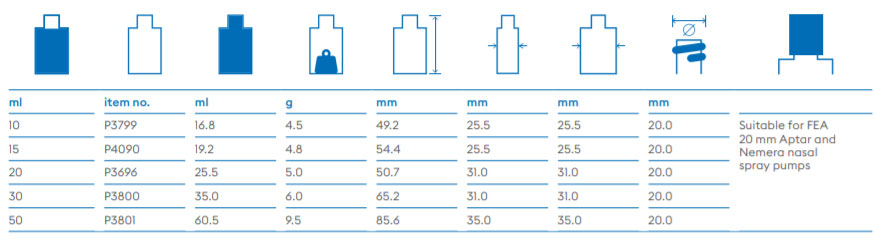
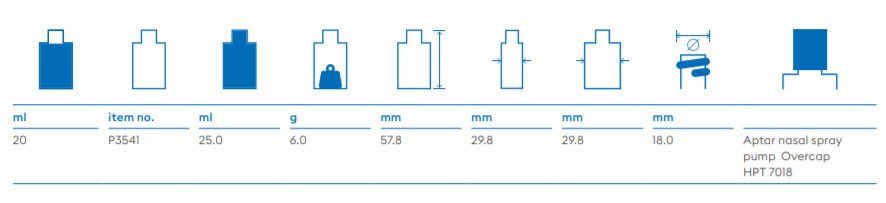
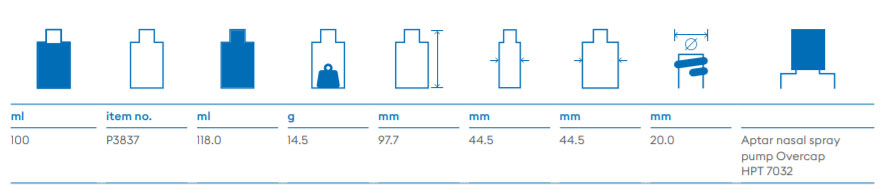
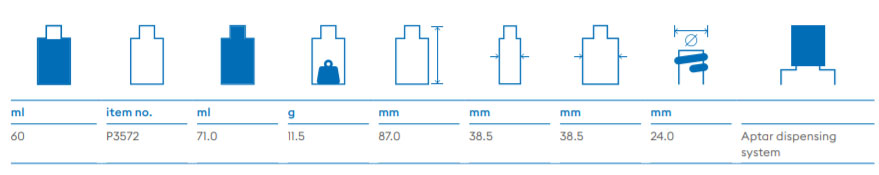
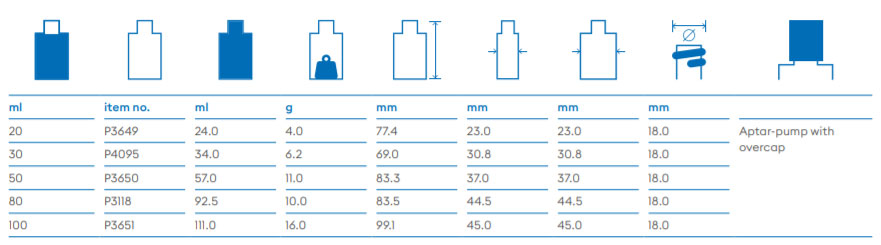
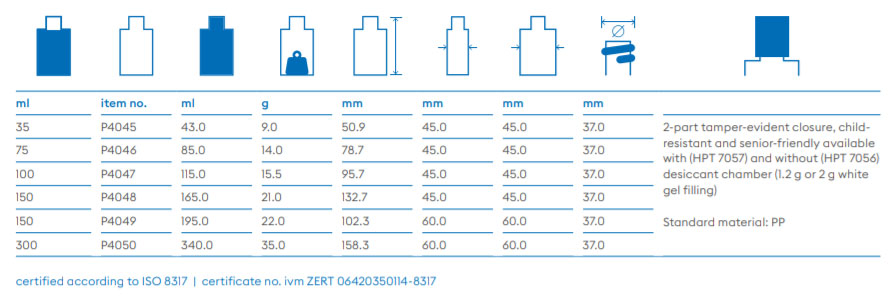
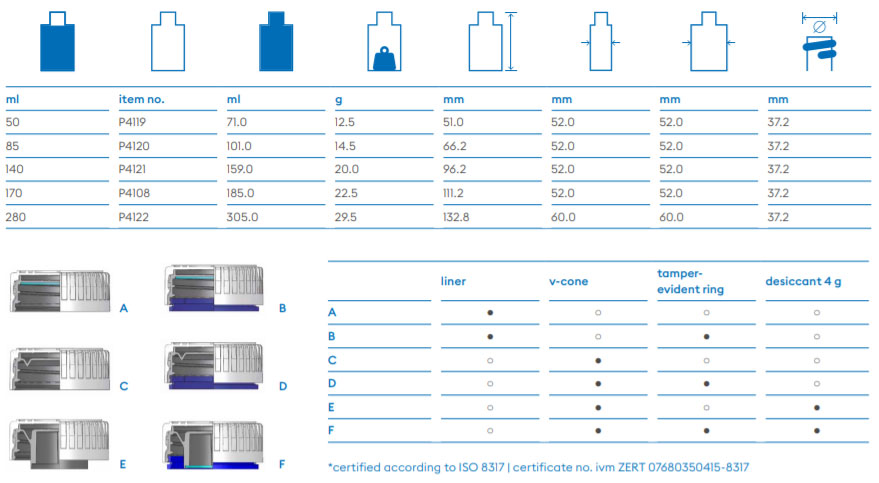
About Us
The Röchling Group has been shaping industry. Worldwide. For nearly 200 years. We transform the lives of people every day with our customized plastics: they reduce the weight of cars, make medication packaging more secure and improve industrial applications. Our workforce of around 11,500 people is located in the places where our customers are – in 90 locations in 25 countries. The Group’s three divisions generated joint annual sales of 2.352 billion euros in 2019.
The Industrial division is the expert for optimal materials for every use. We develop and supply individual products made of plastic for all industrial areas. This is why we have the broadest product range of thermoplastics and composite materials. We supply our customers with semi-finished products or machined components.
The Automotive division advances mobility. Our product solutions in the areas of aerodynamics, propulsion and structural lightweight help solve major challenges. We protect the environment while also improving the driving experience.
The Medical division is the reliable partner to leading global companies when it comes to the components, services and smart plastic products that are needed in the healthcare industry. We develop solutions in the fields of diagnostics, fluid management, pharma as well as surgery and interventional.